
Chemistry, 14.02.2023 14:00 itsmichaelhere1
What is the limiting reagent when 1.5 moles of nitrogen react with 6 moles of hydrogen? N2(g)+3H2(g)—> 2NH3(g)
Answers

Answer from: ulilliareinhart2
Explanation
Given
N2(g)+3H2(g)—> 2NH3(g)
Number of moles of nitrogen = 1.5 moles
Number of moles of hydrogen = 6 moles
Required: Limiting reagent
Solution
For Nitrogen:

For Hydrogen:
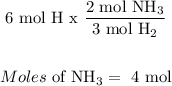
The limiting reagent is Nitrogen, because less moles of NH3 will be produced by nitrogen, meaning it all of it gets used up.
Answer
The limiting reagent is Nitrogen.

Answer from: Quest
it would be a gas formation

Answer from: Quest
The answer is 65.3312
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 19:00, elizabethajih99
Sum of brother and sisters age is 26. four times the brothers age is subtracted from three times the sisters age, the difference is 8. what are the ages of the brother and sister?
Answers: 1

Chemistry, 23.06.2019 01:40, mandilynn22
Calcium carbonate decomposes at high temperatures to give calcium oxide and carbon dioxide as shown below. caco3(s) cao(s) + co2(g) the kp for this reaction is 1.16 at 800°c. a 5.00 l vessel containing 10.0 g of caco3(s) was evacuated to remove the air, sealed, and then heated to 800°c. ignoring the volume occupied by the solid, what will be the mass of the solid in the vessel once equilibrium is reached?
Answers: 1

You know the right answer?
What is the limiting reagent when 1.5 moles of nitrogen react with 6 moles of hydrogen? N2(g)+3H2(g)...
Questions in other subjects:




Mathematics, 19.12.2019 11:31

Physics, 19.12.2019 11:31


Advanced Placement (AP), 19.12.2019 11:31


Mathematics, 19.12.2019 11:31

