
Chemistry, 14.02.2023 14:00 itsmichaelhere1
What is the limiting reagent when 1.5 moles of nitrogen react with 6 moles of hydrogen? N2(g)+3H2(g)—> 2NH3(g)
Answers

Answer from: ulilliareinhart2
Explanation
Given
N2(g)+3H2(g)—> 2NH3(g)
Number of moles of nitrogen = 1.5 moles
Number of moles of hydrogen = 6 moles
Required: Limiting reagent
Solution
For Nitrogen:

For Hydrogen:
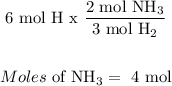
The limiting reagent is Nitrogen, because less moles of NH3 will be produced by nitrogen, meaning it all of it gets used up.
Answer
The limiting reagent is Nitrogen.

Answer from: Quest
The answer is yes b ffgnbsfgnbnfgbnfgsnb

Answer from: Quest
65uuyhjghj8ykitdyidii
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, micvar9646
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 00:30, boonkgang6821
The clouds are grey and ground is wet. a quantitative b qualitative
Answers: 1

Chemistry, 22.06.2019 10:30, cheyennecarrillo14
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 22.06.2019 12:50, martinez6221
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1
You know the right answer?
What is the limiting reagent when 1.5 moles of nitrogen react with 6 moles of hydrogen? N2(g)+3H2(g)...
Questions in other subjects:

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

